
A recent study is further unraveling the mystery surrounding the bluing reaction of psilocybin mushrooms (aka psychedelic mushrooms or magic mushrooms) in the genus Psilocybe.
Blue bruising (from bumping, cutting, or another injury) is a signature feature of psilocybin-containing mushrooms. Since the 1960s, scientists have known that enzymes in mushrooms oxidize psilocin to a blue color.1–5 However, these tests were done using mammalian tissues, and they may not represent the mechanism by which the blue color forms in mushrooms.
Bluing provides strong evidence that a particular mushroom is an active psilocybin mushroom. According to mushroom expert Paul Stamets, blue bruising is one of two features often used for determining if a mushroom has a high probability of containing psilocybin: blue bruising and a purple-brown spore print.6
The Enzymes Involved in Magic Mushroom Bluing
Researchers at the Hans-Knöll Institute in Jena, Germany, used spectroscopy technologies to identify two enzymes in Psilocybe cubensis that lead to blue reaction products.7 The study found that PsiP (a phosphatase) and PsiL (a laccase) degrade psilocybin (Figure 1) in a cascade reaction. This degradation prepares the psilocybin molecule for oxidative oligomerization that leads to the formation of a blue compound. Specifically, PsiP removes the 4-O-phosphate group from psilocybin to form psilocin (Figure 1). At the same time, PsiL oxidizes the 4-hydroxy group.
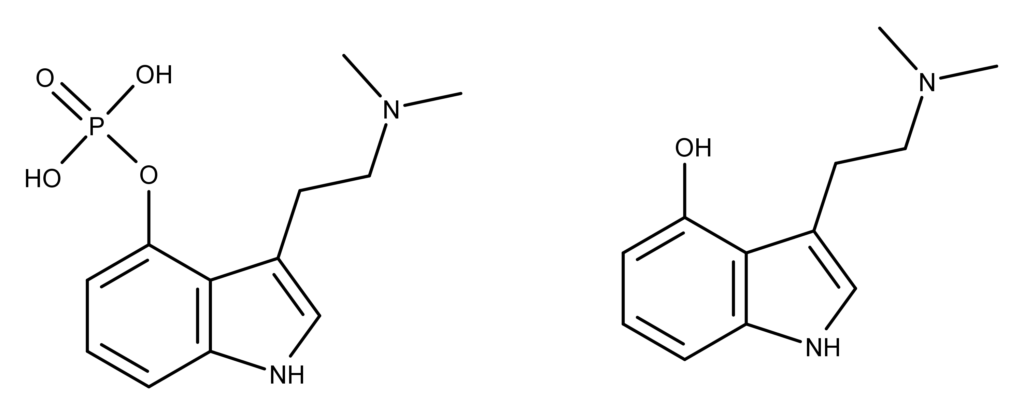
Figure 1: The chemical structures of psilocybin and psilocin. Note that psilocin is dephosphorylated psilocybin, a reaction catalyzed by the enzyme PsiP.7
Dimeric, Trimeric, and Tetrameric Chromophores of Psilocin
Using MALDI-MS (matrix-assisted laser desorption/ionization mass spectroscopy) and in situ NMR (nuclear magnetic resonance) spectroscopy, three chemical structures of the chromophores responsible for bluing were identified. The main structure is shown in Figure 2. The study authors summed up their findings with the statement,
“…the blue color is due to a heterogeneous mixture of quinoid psilocyl oligomers, primarily coupled via C-5.
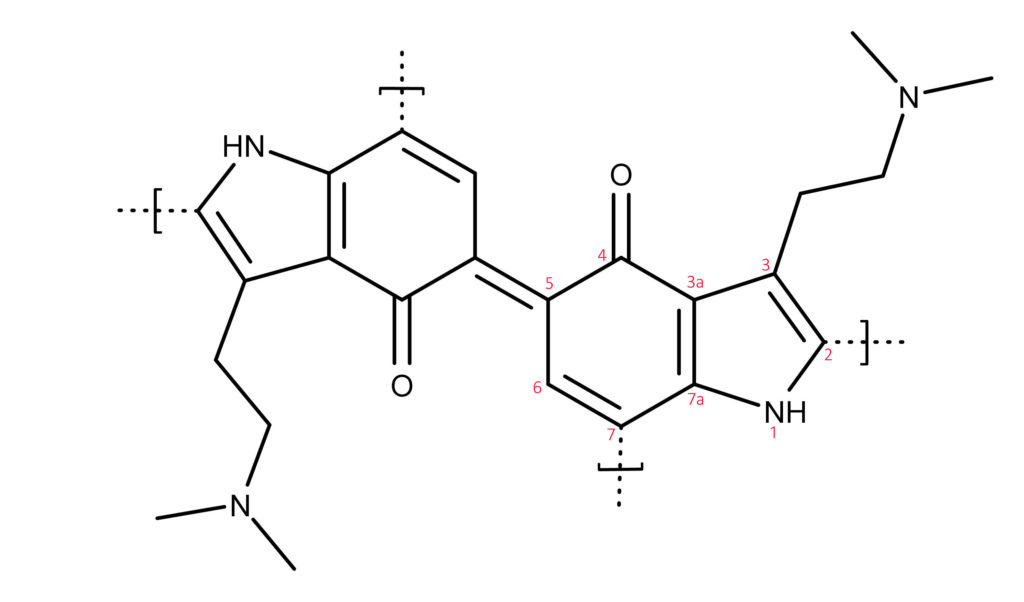
Figure 2: The chemical structure of the primary quinol compound that makes magic mushrooms turn blue.7 This basic structure is two psilocin molecules (called a dimer) connected by a double bond at the 5th position. The quinoid results when the -OH group on carbon 4 is oxidized to =O by the enzyme PsiL. The dotted lines with brackets indicate additional carbons where oxidized psilocin can attach to create a quinoid trimer. A quinol tetramer is formed when oxidized psilocin attaches to all four carbons. The oxidation reactions are reversible.
Dr. Dirk Hoffmeister of the Leibniz Institute for Natural Product Research and Infection Biology at the Hans-Knöll Institute and his research team have been working with magic P. cubensis for years. During that time, they have curiously witnessed the bluing phenomenon and set their sights on solving the mystery. However, their interest in magic mushrooms is not just about their chemistry. In a recent interview, Dr. Hoffmeister told Chemistry World,
Psilocybin is looked at as this illegal, recreational drug, but it has a fantastic potential as a medication for therapy resistant depression.
There’s More to Learn About the Chemistry of Magic Mushroom Bluing
This study data indicates that the blue (or in some cases blue-green) color observed in magic mushrooms may be due to one or more combinations of compounds, collectively “bluing compounds.” These bluing compounds can exist as dimeric, trimeric, and tetrameric forms of psilocin and/or its structural analogs. The multitude of different bluing compounds explains the different shades of blue and blue/green that are observed in different species of magic mushrooms.
There may be several biosynthetic routes by which the mushrooms created bluing compounds. Based on the data from this study, the oxidative power and substrate concentration appear to be limiting factors in how much of each oligomer is present. More research will help clarify these aspects of the bluing mechanism.

Could there be some psychedelically active mushrooms that don’t bruise blue for lack of PsiP (a phosphatase) and PsiL (a laccase)?
Blue bruising boletes are generally toxic
One example of a species that is considered quite potent but only has minimal bluing (and sometimes no bluing) is the libery cap mushroom (Psilocybe semilanceata). It would be very interesting if scientists could quantify the levels of the “bluing enzymes” (PsiP and PsiL) in different species of mushrooms for comparison.
I have the same question are there suicide mushrooms magic mushrooms that do not bruise blue or show signs of bluing but have spore prints matching and all other indicators leading to identification besides the bluing.
Yes. There are panelolus that only contains psilosin so they do not bruise blue and some psilocybe also and a couple mycena and others. The bluing is the psilocybe degredating to psilosin I don’t think this will affect the strength though as when we do lemon Tek or other teks to convert to psilosin it also makes it blue as fuck. It is a chemical change from psiocyben that causes the blue and psilosin by itself will not bruise blue but when psilosin is taken instead of psilocybe either by crushing or Tek,we do this to save the sick feeling… Read more »
Muchas gracias
Could microdosing cause deranged bloods and liver toxicity in someone with SBS ? And increased copper and iron in bloods ?