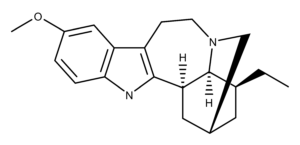
Ibogaine is an alkaloid compound that was first isolated from the plant Tabernanthe iboga that grows in Central Africa.1 Specifically, the plant is a shrub also known as iboga.
Traditionally, iboga plants are used in cultural and religious rituals by the indigenous peoples of Central Africa.2 They also use it in various dilutions for alleviating hunger, fatigue, and thirst.
The Chemistry of Ibogaine
Ibogaine was first isolated by Dybowski and Landrin in 1901.1 Its crystal structure was solved in 1960.3 The first synthesis by done in 19664 and a simplified total synthesis was published in 2012.5 A detailed summary of ibogaine synthesis is found in Wasko et al., 2018.6
Ibogaine crystallizes into prismatic needles from ethanol.7 In addition to ethanol, it is soluble in ether, chloroform, acetone, and benzene. It is practically insoluble in water. The hydrochloride salt of ibogaine is soluble in water, as well as methanol and ethanol. It is slightly soluble in acetone and chloroform, but practically insoluble in ether.
The Pharmacology of Ibogaine
Ibogaine is cardiotoxic at micromolar levels.8–11 Specifically, studies have noted a prolongation of the heart’s QTc interval. Ibogaine has also been shown to be neurotoxic in rodents.12 At higher doses, the effects include tremors, convulsions, nervous behavior, and paralysis of the limbs.13 However, the doses used in these studies are below what would be used in a clinical setting.8
In terms of pharmacodynamics, ibogaine shows no clear preferences, having a moderate to weak affinity for a variety of receptors and transport proteins (see summary in Wasko et al. 2018). As a result, the hallucinogenic effects of ibogaine cannot be attributed to the activation of the serotonin 5-HT2A receptor (Ki = 16 µM).12 However, ibogaine’s principal metabolite noribogaine has sub-micromolar affinity (0.61 µM) as a partial agonist of the kappa opioid receptor.14 The mechanism of the dissociative effects caused by ibogaine may be similar to that of ketamine and other NMDA (N-methyl-D-aspartate) channel blockers.15,16
Ibogaine also binds in the low micromolar range to the mu opioid receptor, as does noribogaine in sub-micromolar range.17,18 This may explain the ability of ibogaine to decrease self-administration of morphine in rats.19,20
Studies indicate that ibogaine’s anti-opiate effects may be due to its noncompetitive antagonist action at nicotinic acetylcholine receptor subtypes including α1ß1 and α3ß4.21,22
The Applications and Potential of Ibogaine
In the early 1960’s, subjective reports of the anti- addictive properties of ibogaine began surfacing, most notably from a young heroin addict named Howard Lotsof.23 Lotsof was born in the Bronx, New York in 1943 and was severely addicted to heroin by the time he was 19 years old. By his own account, the desire of he and six of his friends to take heroin disappeared almost immediately after self-administration of an extract of the T. iboga root. Lotsof became one of the most vocal advocates of ibogaine use for the treatment of addiction. In 1985, he was granted a patent for the treatment of cocaine and heroin addiction using ibogaine.24
Research indicates that ibogaine may be effective in treating opiate addiction.25–28 The data show that it doesn’t just reduce opioid use, but can result in complete abstinence, along with having “long-term positive psychological outcomes.”28 A 2018 study found that the positive effects of ibogaine for treating opioid dependence lasted twelve months.25 In a 2018 review paper, Deborah Mash et al. recommended using a single oral dose of ibogaine during detoxification “to transition drug dependent individuals to abstinence.” 26