The serotonin 5-HT2A receptor is important for researchers studying psychedelic drugs. Little is known about if and how the other serotonin receptor subtypes are involved in the overall psychedelic experience. Another serotonin receptor subtype, 5-HT3, is of current interest to researchers designing drugs to help cancer patients who experience nausea and vomiting during chemotherapy. Even if 5-HT3 is not involved in the psychedelic experience, it’s interesting to examine the work being done by researchers in this area.
The Basics of Serotonin
Serotonin (also known as 5-hydroxytryptamine or 5-HT) is an important pharmaceutical target.1 It is a neurotransmitter and regulator thought to be responsible for feelings of happiness and well-being in humans. Serotonin (Figure 1) has other essential functions in the body including digestion, blood clotting, and bone metabolism. All mammals make and use serotonin, as well as bilateral animals including insects.
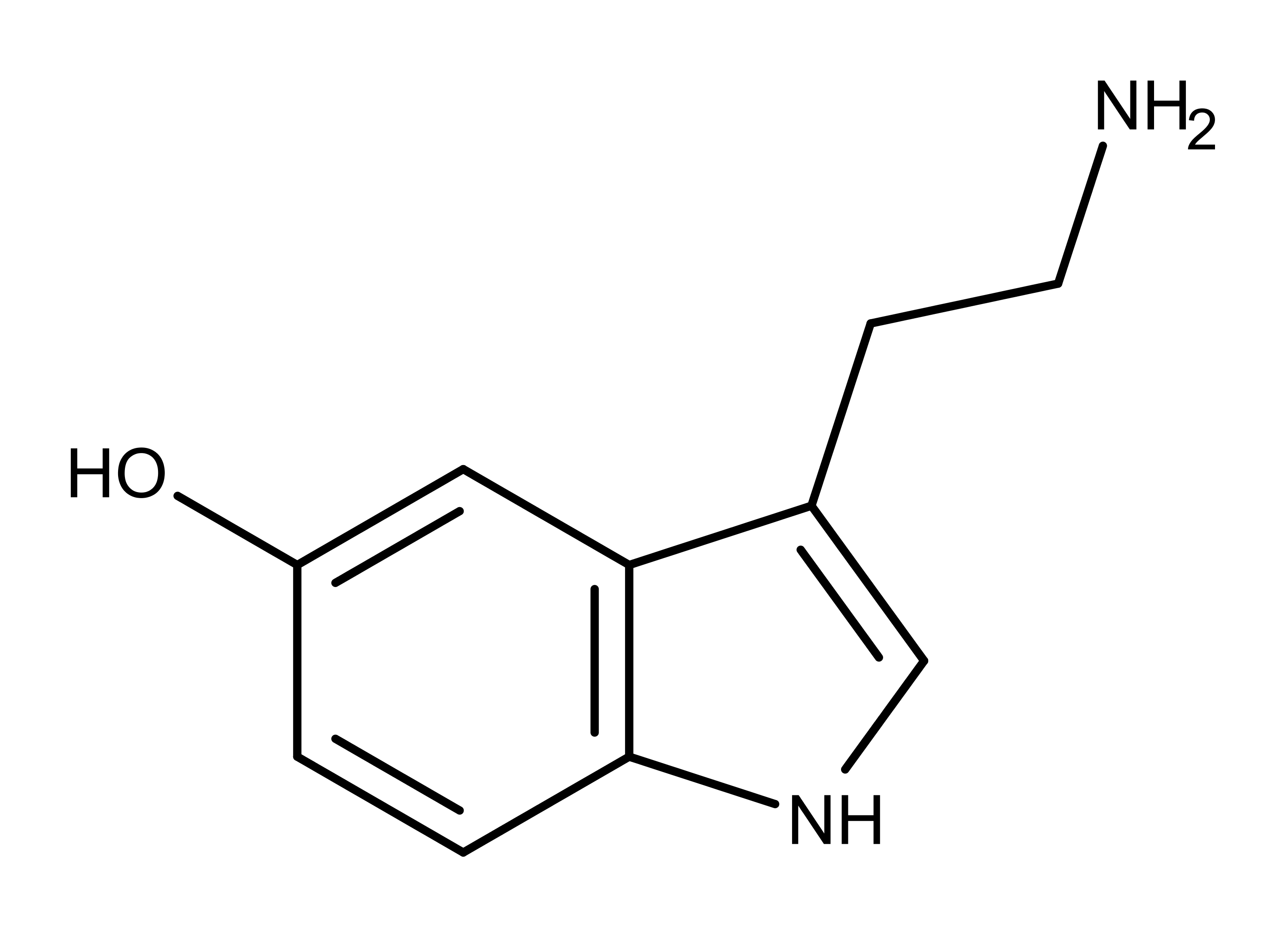
Figure 1: The chemical structure of serotonin.
Modulating the release of other neurotransmitters such as dopamine, epinephrine, norepinephrine, acetylcholine, and glutamate is another function of serotonin. Also, many hormones are regulated by serotonin including oxytocin, cortisol, vasopressin, and prolactin. Various neurological and biological processes like aggression, appetite, anxiety, sleep, nausea, mood, memory, and thermoregulation are influenced by serotonin as well.
Serotonin is synthesized from the essential amino acid tryptophan. The pathway is controlled by the enzyme tryptophan hydroxylase-2.2 The body stores serotonin serotonergic neurons in the central nervous system (CNS) and intestines, the enterochromaffin cells in the mucosa of the gastrointestinal (GI) tract, and in blood platelets.
The Serotonin Receptors
The action of serotonin is mediated through receptors found throughout the CNS and peripheral nervous system (PNS). Activation of the receptors (when they bind serotonin) causes excitatory or inhibitory neurotransmissions. There are seven main categories of serotonin receptors in humans (5-HT1-7) with 14 known types.3 Receptors with subscripts containing A, B, C, etc. are known as subtypes.
5-HT1A
5-HT1B
5-HT1D
5-HT1E
5-HT1F
5-HT2A
5-HT2B
5-HT2C
5-HT3
5-HT4
5-HT5A
5-HT5B
5-HT6
5-HT7
These receptors are located all over the body. Some are concentrated in areas more than others. For example, 5-HT1F and 5-HT6 are found almost exclusively in the CNS while 5-HT2A is more ubiquitous in blood vessels, CNS, PNS, GI tract, platelets, and smooth muscle.
Drugs that bind to serotonin receptors can be agonists, inverse agonists, or antagonists. When an agonist like serotonin binds to a receptor, it produces a biological response. Antagonists block the action of agonists while inverse agonists produce a response that is opposite of the agonist. Also, there are different degrees of binding. For example, a partial agonist gives only a partial effect compared to a full agonist.
Serotonin receptors are the targets of antidepressant drugs known as selective serotonin reuptake inhibitors or SSRIs. However, in the case of most SSRI drugs, this is only a theory and the exact mechanism is not fully understood.
Research and Development is Driven by Need
Little research work has been done developing 5-HT3 agonists. Pharmaceutical companies that have expertise with the 5-HT3 receptor are focusing on receptor antagonists, also called antiemetics.4 This is because activation of the 5-HT3 receptor causes nausea and vomiting which are common side effects of chemotherapy and radiation treatments. In spite of health programs and increased awareness, cancer diagnoses continue to increase around the world.
In 2018, there have been an estimated 18 million cases of cancer globally.5 The number is expected to reach 21.7 million by 2030.6 This will increase the use of chemotherapeutic agents for treatment leading to nausea and vomiting in a growing number of patients. Research and development into anti-nausea 5-HT3 receptor antagonists such as ondansetron, granisetron, and dolasetron7 will far surpass research work on agonists because of the ever increasing need.
State of the Art of 5-HT3 Agonists
A data sampling taken from ClinicalTrials.gov reveals how few pharmaceutical companies and researchers are currently working on agonist drugs targeting the 5-HT3 receptor. Tables 1 and 2 show the top results.
Table 1: Companies researching 5-HT3 receptor drugs based on the number of studies in the literature.
| Company | Number of Studies |
| H. Lundbeck A/S | 37 |
| Takeda | 36 |
| Merck Sharp & Dohme Corp. | 3 |
| Takeda Pharmaceuticals North America, Inc. | 2 |
| ElMindA Ltd | 1 |
| Lundbeck Canada Inc. | 1 |
Table 2: Universities and organizations researching 5-HT3 receptor drugs based on the number of studies in the literature.
| University/Organization | Number of Studies |
| Peking University First Hospital | 3 |
| Affiliated Hospital of Hebei University | 2 |
| Beijing Chao Yang Hospital | 2 |
| Beijing Ditan Hospital | 2 |
| Beijing Shijitan Hospital | 2 |
| Beijing Tsinghua Chang Gung Hospital | 2 |
| Guizhou Provincial People’s Hospital | 2 |
| Massachusetts General Hospital | 2 |
| Peking University International Hospital | 2 |
| Qingdao Municipal Hospital | 2 |
The 5-HT3 receptor is unique compared to other serotonin receptors.8 The other receptors function by means of G-protein coupling (they detect molecules outside the cell and activate internal signaling pathways). 5-HT3 is a ligand-gated Na+ and K+ cation channel. In other words, when a ligand like serotonin binds to the receptor, it opens ion channels in the cell membrane allowing combinations of ions like Na+, K+, Ca2+, and Cl– to flow in and out. Depending on what flows in and out and how much, the ligand binding triggers depolarization (excitatory response) or hyperpolarization (inhibitory response) of the plasma membrane of the cell.
The biochemical structure of the 5-HT3 receptor began to emerge in 20138 and the first x-ray crystalline structure for the mouse receptor was captured in 2014.9 The receptor binding and gating mechanisms were further described by researchers in 2016.10,11
Although it’s always a dynamic situation, online references show about a dozen 5-HT3 agonists in the drug market or being used in research.
- Cereulide – A toxin produced by the bacteria Bacillus cereus.
- 2-methyl-5-HT – A derivative of tryptamine.
- Alpha-methyltryptamine – A psychedelic stimulant marketed briefly in Russia as Indopan.
- Chlorophenylbiguanide – An allosteric agonist/modulator of 5-HT3 and antagonist of α2A-andrenergic receptor.
- Ethanol – Also known as ethyl alcohol or grain alcohol.
- Ibogaine – A naturally-occurring psychoactive found in the plant family Apocynaceae.
- Phenylbiguanide – A molecule used in research to study the function of the 5-HT3
- Quipazine – Used in research. Also binds to 5-HT2A.
- RS-56812 – A potent and selective partial agonist. Improves memory in animal tests.
- SR-57227 – A potent and selective partial agonist. Penetrates the blood-brain barrier.
- Varenicline – A prescription medication for treating nicotine addiction.
- YM-31636 – A 5-HT3 agonist that improves colonic motility (constipation) in rats.
Toad Venom
In addition, several species of toads in the genus Bufo have venom in their skin and eggs which contains bufotenin (also spelled bufotenine) chemically known as N,N-Dimethyl-5-hydroxytryptamine or 5-HO-DMT.12 This 5-HT3 agonist is also found in mammals, mushrooms, and higher plants. Interestingly, it is often detected in the brains, urine, and plasma of people with schizophrenia. 5-HO-DMT is a psychedelic structural analog (their chemical structures are similar) of serotonin, psilocybin, and DMT. Recently, researchers have identified bufotenin as a candidate for treating rabies.13
Bufotenin is chemically related to bufotenidine or 5-hydroxy-N,N,N-trimethyltryptammonium.14 This is a 5-HT3 agonist used in research to study how the 5-HT3 receptor works. It has research limitations, however, due to the quaternary amine group which prevents it from crossing the blood-brain barrier.
More Research is Needed
Recent studies indicate 5-HT3 may be a therapeutic target for epilepsy drugs,15 obsessive-compulsive disorder,16 and possibly antidepressants.17 In spite of everything that is known, a cloud of mystery surrounds the 5-HT3 receptor and the others in the serotonin receptor family. Even more intriguing is how these receptors work in concert to create and modulate their effects. The vast majority of this interaction is unknown and not being studied by scientists. The therapeutic implications of understanding how these receptors work individually and together cannot be overstated.
