
Background
Lysergic acid diethylamide (LSD) was the first major psychedelic substance of entirely synthetic origin and became an early mainstay of psychiatric medicine.1 While the substance was eventually criminalized across the world by the 1970s, medical interest in LSD has resurged in the last decade, spurred on by positive results for another hallucinogen, psilocybin, in recent clinical trials focusing on mental health disorders.2
Interest has also renewed in other effects of LSD, especially its anti-inflammatory properties. The endocannabinoids are a group of fatty acid neurotransmitters that act on the cannabinoid receptors and are a major contributor to resolving inflammation in humans. It is possible that the anti-inflammatory effects of LSD are mediated through endocannabinoids. The weight of evidence has also placed endocannabinoids as a centerpoint in communication between the gut microbiome and human metabolism and immune function.3
In a new paper, Inserra and colleagues used an animal model to study the pro-social effects of LSD and identify any relationship with endocannabinoid and serotonin neurotransmitters, as well as changes to the gut microbiome.4
Study design
The authors used a mouse model to study changes in social behaviour in response to semi-chronic dosage of LSD (Figure 1). Mice in the treatment groups were given 30 μg/kg/day by injection for 7 days, while those in the control group were administered a placebo. After dosing, animals were put into two behavioral tests. Fecal pellets were collected across the study to test the composition of the gut microbiome at various time points.
At the end of the behavior studies, the quantity of endocannabinoids and related compounds in the LSD-treated mice were determined by liquid chromatography tandem mass spectrometry, the gold standard technique.
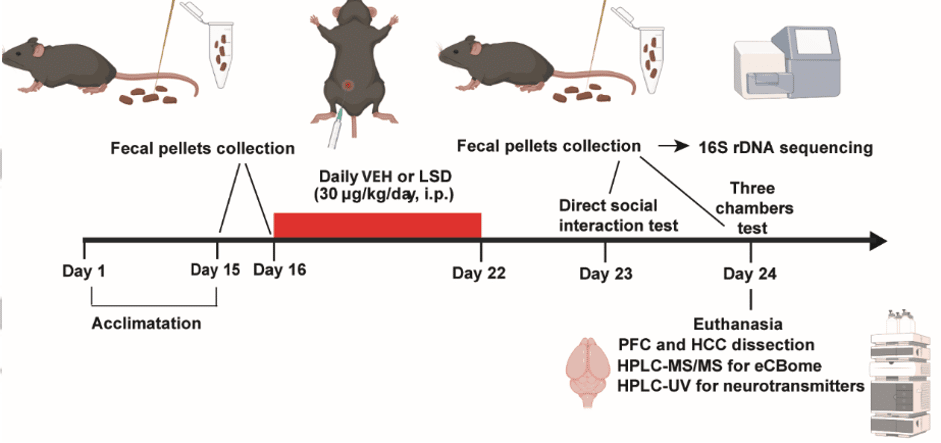
Figure 1: Graphic overview of in vivo experiment design. After a period of acclimation, mice were treated with either LSD injection or vehicle control for 7 days. After this chronic treatment, experimental behaviour studies were performed. Throughout the study, samples were taken for analysis. Adapted from Inserra et al., 2022 by Kieran Deane-Alder.4
LSD enhances social interaction
The first test, which examined direct social interaction (DSI) between a mouse in its “home” territory, and an “invader” mouse it had not met before, showed that LSD-treated mice engaged in around 50% more interactions with the stranger than did the control mice.
In an additional experiment, mice were offered the choice between staying put in a familiar cage, exploring an empty cage, or interacting with two “stranger” mice (the second stranger introduced after a delay). While both LSD-treated and control mice were more interested in the strangers than the empty cage, in keeping with the direct interaction results, LSD-treated mice spent even more time interacting with their new friends. Furthermore, LSD-treated mice showed a greater preference for spending time with the second stranger mouse, suggesting they were more engaged by novel stimuli.
The researchers also measured other common behaviors in the mice – rearing, grooming, and total movement. These were not affected in the LSD-treated mice, giving more confidence that the increased interaction between LSD-treated mice and the “stranger” mice could be attributed to prosocial enhancement by the psychedelic drug.
LSD-prompted changes in endocannabinoids in the hippocampus correlated with pro-social behavior
Curiously, the effect of LSD treatment on the brain endocannabinoidome (eCBome) – the network of endocannabinoid precursors, major active molecules, and their metabolites – was limited to the hippocampus. Endocannabinoids are modified fatty acids that act as neurotransmitters, and the two major compounds, anandamide and 2-arachidonoyl glycerol (2-AG), act directly on cannabinoid receptors and are both based on the fatty acid arachidonic acid (Figure 2).
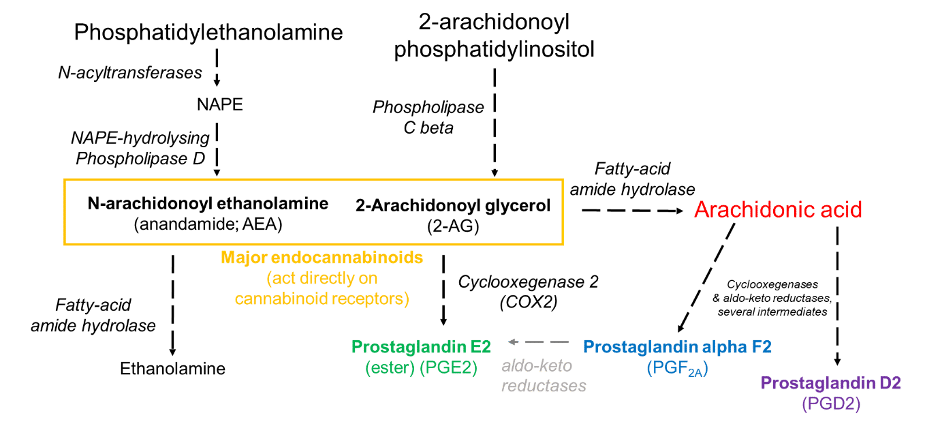
Figure 2: Simplified schematic of endocannabinoid metabolism. Compounds with major roles in inflammation are coloured. Enzymes are italicised. Minor routes in grey. Analogous routes with different enzymes produce endocannabinoid-like substances (i.e. N-acylethanolamines and monoacylglycerols) such as N-linoleoylethanolamine (LEA; containing linoleic acid, instead of arachidonic acid as for anandamide). Created by Kieran Deane-Alder.
In this study, the amount of anandamide in the tissue was reduced in LSD-treated mice, as were N-linoleoylethanolamine (LEA) and docosahexaenoylethanolamine (DHEA), which are similar compounds to anandamide based on different fatty acids. The endocannabinoid-like 1/2-docosahexaenoylglycerol (1/2-DHG), similar to 2-AG, was substantially decreased. Also decreased were levels of important inflammatory mediators prostaglandin D2 (PGD2), prostaglandin alpha F2 (PGAF2), and thromboxane 2 (TXB2). These changes correlated well with enhanced social behavior from the same animals earlier in this study. This was not expected, as increases in endocannabinoid levels have consistently been positively linked to social behavior, as noted by the authors; however, it was also recently established that d9-tetrahydrocannabinol (THC), which acts similarly to endocannabinoids in the brain, can indeed diminish sociability in mice.5
Changes to tryptophan metabolism
The authors also looked at differences in tryptophan metabolism in a separate cohort of LSD-treated mice. This may be of significance as LSD targets serotonin (5-HT) receptors in the brain, and serotonin is mainly synthesized from tryptophan. The authors found that levels of kynurenine, a metabolite of tryptophan that has been linked to inflammation and depressive symptoms in humans, were decreased in LSD-treated mice.
LSD had an equivocal impact on the mouse microbiome
Overall, the composition of the gut microbiome was not substantially different in LSD-treated mice; however, the overall diversity of microbes was reduced. Considering changes over the study’s timeline, vehicle-treated mice had an increase in the ratio between Firmicutes and Bacteroidetes species (proposed as an indicator of a “disrupted” microbiome), whereas the LSD-treated mice did not. This could indicate a protective effect from LSD.
Conclusion
The authors found reductions in endocannabinoid-related inflammatory signalling molecules in the brains of mice treated with LSD. The localization to the hippocampus was not anticipated and is an interesting observation. Also unexpected was the correlation between social behaviour and a reduction in endocannabinoids. It will be interesting to see if these findings are repeated and if this is an effect specific to LSD, or to classical psychedelics more generally.
