In a March 2020 Psychedelic Science Review article, Dr. Alexander Sherwood of the Usona Institute noted that psychedelic research had been impeded by a lack of access to pure, well-characterized research chemicals. The article also covered his addressing this unmet need by publishing synthetic methods for making several psilocybin analogs.1
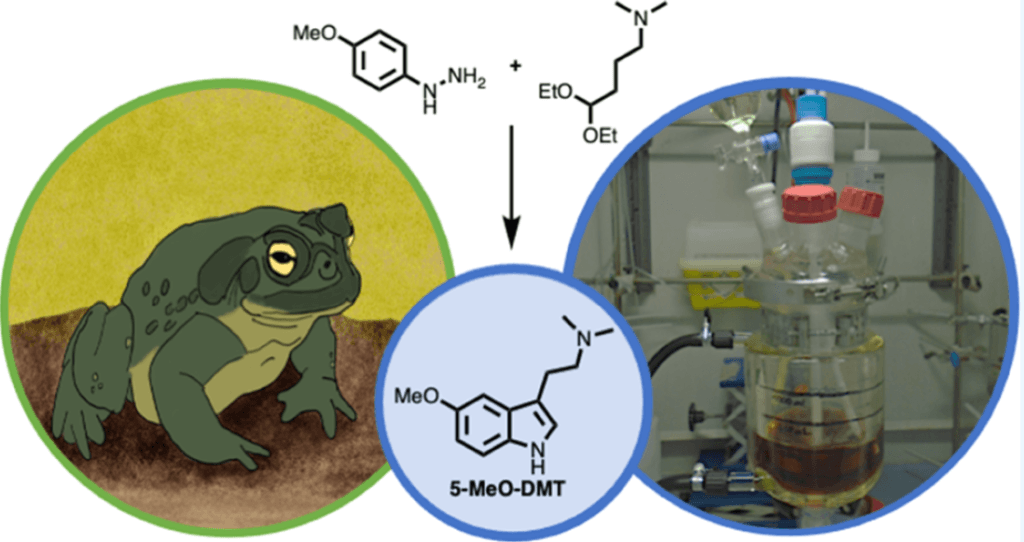
Dr. Sherwood is the lead author of a recently published paper in ACS Omega titled “Synthesis and Characterization of 5-MeO-DMT Succinate for Clinical Use.” 2 The paper details a synthetic route for making the succinate salt of 5-MeO-DMT, a psychotropic compound found in the venom of some toad species. This effort is another step forward in providing pure, well-characterized research chemicals for use in psychedelic research.
In the paper, Dr. Sherwood explains the critical nature of the work. Currently, there are only a handful of psychedelic compounds in clinical development, and improving access to pure compounds would enable the needed research studies and clinical trials.
Why Make the Succinate Salt of 5-MeO-DMT?
The paper explains in detail the rationale behind the research team’s decision to focus on 5-MeO-DMT succinate. First, they examined the available evidence to determine the best route of administration for the compound. They found chemical, physiological, and practical drawbacks to methods such as smoking, vaporizing, transdermal, and intravenous administration.
They followed the research to a 2020 study published by Malin Uthaug et al.3 This study reported that the intramuscular (IM) injection of 5-MeO-DMT in a naturalistic setting was “…associated with lower and less doses, lower frequencies of reporting reactivation, a higher frequency of physical tension release, and longer onset of acute effects” compared to vaporization. Sherwood et al. noted that IM injection also allows for precise metering of doses.
Working from a 2018 study by Gupta et al.4 the team stated that “…a range of pharmaceutically acceptable salt forms were considered from acids with sufficient pKa difference to fully protonate 6 [5-MeO-DMT], including the counterions chloride, sulfate, fumarate, succinate, maleate, lysate, oxalate, benzoate, tartrate, mesylate, or acetate.” Testing narrowed the field down to four salt forms: hydrochloride, sulfate, fumarate, and succinate.
Various issues with water solubility and chemical stability were identified with three of the salts. The succinate form was chosen due to its high water solubility and having less potential for chemical reactivity. The latter is especially important during autoclave sterilization, which may be required for preparing sterile solutions.
5-MeO-DMT Succinate Synthesis Overview
As part of this study, Sherwood et al. developed an improved method for synthesizing 5-MeO-DMT freebase, the starting material for making the succinate salt. They explained, “The key features of the developed process were an optimized Fischer indole reaction with advantageous inclusion of acetonitrile cosolvent to provide crude freebase.” Also noted were “…greener solvent choices with an intermediate purification via filtration through a silica pad.”
Their new synthesis method for making the 5-MeO-DMT succinate API (active pharmaceutical ingredient) resulted in a 49% overall yield and 99.86% purity of the final product. The method includes the use of methanol, activated charcoal decolorizing, and purification using an acetone slurry.
Along with the succinate acid salt, the team identified a degradation product, the N-oxide form of 5-MeO-DMT. However, research by Shen et al. in 2010 indicates that the N-oxide form is also a 5-MeO-DMT metabolite.5 As such, Sherwood et al. explained that this allows some flexibility in how much N-oxide can be in the API.
In concluding, Sherwood et al. stated, “The first production run has provided sufficient API to meet current clinical and nonclinical needs to enable first-in-human clinical trials with 6 [5-MeO-DMT].”
Understanding Psychedelic Drug Pharmacology is Critical to Psychedelic Therapies
Interestingly, Dr. Sherwood notes that psychedelic drugs are generally non-selective, “simultaneously interacting” with several serotonin receptors in a “synergistic way,” resulting in “variable degrees of synergistic polypharmacology.” Given this breadth and variability of psychedelic pharmacology, it is feasible that various psychedelic compounds could be used within different contexts or for treating different mental and physical conditions.
Until psychedelic science reaches the level of understanding necessary for making precise formulations, it seems that one course of action is making and studying a wide range of psychedelic compounds. This recent paper by Sherwood et al. serves the goal of providing access to another compound (5-MeO-DMT) with significant clinical potential.

I would love to be part of this study. This is would be great for some mental, and physical conditions that I have.
What is one to think of the online sites that offer “research” chemicals such as 5-MeO-DMT?
Yes yes yes there are benefits to all these wonderful, naturally occurring psychedelics (though I do get on a larger scale why they want to make them pharmaceutical grade) potency, etc, I’m just SO VERY glad that things look like they are starting to take a major turn in mental health. It may be slow, but I really believe that we are going to start seeing suicide rates go down when we can get things such as ketamine, psilocybin/psilocin, LSD, and other hallucinagions mainstream. Ketamine made a HUGE difference in my suicidal thoughts. I was blown away as have others!!!