
Drawbacks of MDMA and the Development of Novel Entactogens
Beginning in the late 1980s, researchers began to uncover evidence of MDMA’s long-term adverse effects including potential damage to dopamine and serotonin neurons in the brain, often termed “neurotoxicity”.1 Subsequent research over the following decades generated a large body of evidence demonstrating neurotoxic effects of MDMA that depended on a variety of factors including body temperature, dose, frequency, and context of use.2–4 MDMA’s neurotoxic effects continue to be a source of controversy in the scientific community, divided on whether appreciable toxicity occurs in humans.
The revelation that MDMA may be damaging to neurons led to the development of numerous related drugs in an effort to identify compounds that retained its core therapeutic effects without subsequent neurotoxicity.5 One such series of compounds developed as presumed non-neurotoxic alternatives to MDMA were the Aminopropylbenzofurans (APBs). The APBs share nearly identical chemical structures with MDA and MDMA except for a slight alteration in which an oxygen atom of the methylenedioxy ring is substituted for a carbon (Figure 1A), generating a benzofuran (Figure 1B). This minor modification of the MDA/MDMA chemical structure gave rise to the APB family, which includes such drugs as 6-APB, 5-APB, 6-MAPB, and 5-MAPB, among others (Figure 1).
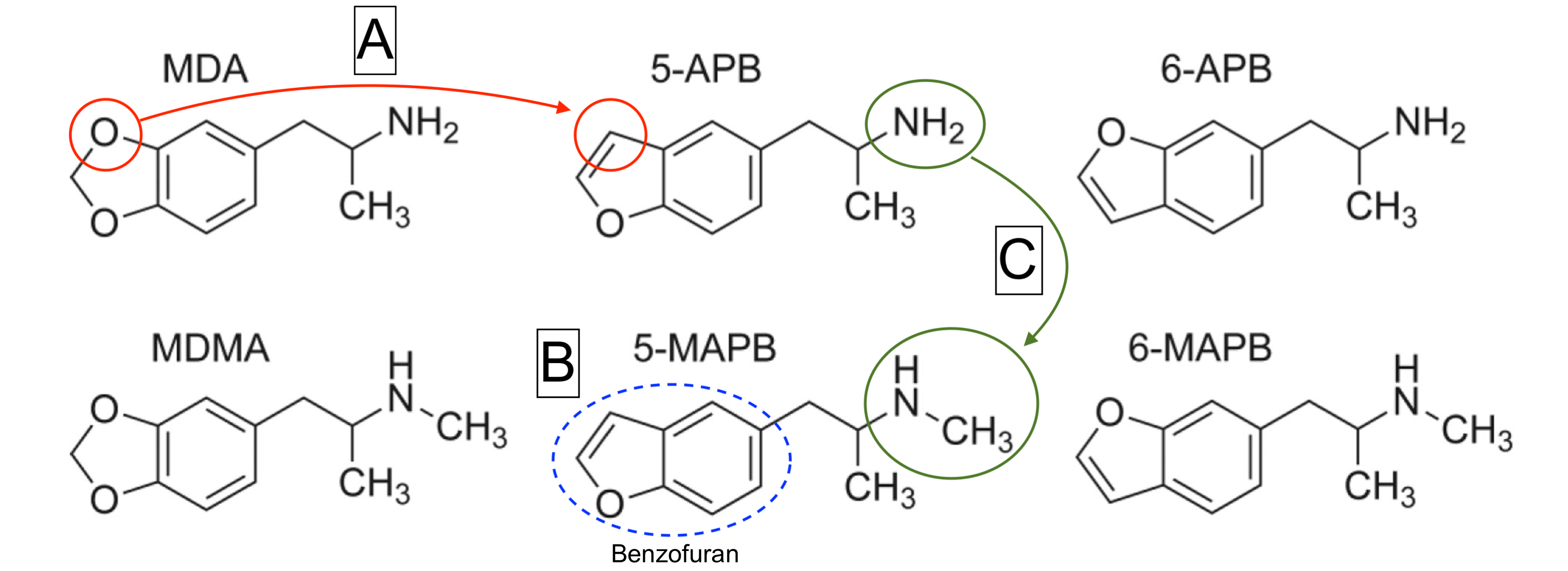
Figure 1: Chemical structures of the prototypical entactogens MDA and MDMA alongside the chemical structures of the most common substituted benzofurans: 5-APB, 6-APB, 5-MAPB, and 6-MAPB. Removal of one oxygen from the methylenedioxy ring and subsequent substitution with a carbon (A) creates a benzofuran structure (B). N-methylation: addition of a methyl group to the nitrogen of 5-APB (C). Adapted from Brandt et al., 2020 by Harrison Elder.
APB Pharmacology
The APBs, due to their structural similarity to MDA and MDMA, act in much the same way as the aforementioned prototypical entactogens.6–8 Pharmacologically, APBs have been shown to act as amphetamine-type releasers of the neurotransmitters dopamine (DA), norepinephrine (NE), and serotonin (5-hydroxytryptamine or 5-HT), as well as agonists at a subset of serotonin receptors including 5HT2A, 5HT2B, and 5HT1A; all mechanisms they share with MDMA and MDA. However, the APBs present slightly different profiles than either of the more well-known entactogens.
Specifically, most APBs are of generally higher potency (e.g. affinity for SERT; 5-APB = 290 nM vs MDMA = 2400 nM), more selective for serotonin release (e.g. DAT/SERT ratio: 5-APB = 0.05 vs MDMA = 0.14), and have different agonist profiles at serotonin receptors (specifically higher affinity and efficacy at 5HT1A, 5HT2A, 5HT2B, and 5HT2C receptors).9–11 They also display altered pharmacokinetic profiles, leading to a different time-course of subjective effects than MDA or MDMA.8,12 In general, phenomenological reports suggest that N-methylated APBs (e.g. 5-MAPB; Figure 1C) have effects similar to MDMA, characterized by a rapid onset and 4-6 hour duration, while APBs containing a primary amine (e.g. 6-APB) are closer in character to MDA, with a protracted onset and longer duration of 7-9+ hours. Despite the moderate differences in pharmacology and effects of these two chemical families, their high degree of similarity led to the emergence of APBs as “designer drug” substitutes for the more common entactogens.
Reports from internet user forums and a small number of publications confirm that the APBs produce core entactogenic effects such as increased sociability, heightened empathy, enhanced emotional bonding, decreased inhibitions, and euphoria at doses that are similar to those required by MDA and MDMA.13 Formal studies were also conducted in which animals are trained to recognize drugs with similar subjective effects. These have shown that APBs are recognized by animals as being MDMA-like, further supporting the similarity of their psychoactive effects.14 Due to the international status of MDMA and MDA as highly controlled schedule I drugs, users and underground psychotherapists have increasingly turned to the gray market for alternatives, leading to the growing popularity of 6-APB, 5-APB, and 5-MAPB as novel entactogens over the past decade.15
The Future of Entactogens
Despite MDMA’s impressive performance in recent clinical trials for PTSD, there is still significant room for improvement of its therapeutic and safety profile. Specifically, the cardiovascular, psychotropic, and possible neurotoxic effects of MDMA are cause for concern in certain patient populations where cardiovascular function, psychological stability, and neurological function may be compromised. While little formal research on the clinical effects of APBs has been conducted to-date, user reports suggest they may be less stimulating, less psychologically taxing, not as prone to induce tolerance, and less likely to cause an unpleasant “crash” after use than MDMA or MDA.8 The neurotoxicity of APBs needs investigating, though the absence of severe post-acute distress and diminished tolerance may be indicative of improvements in this domain as well. Conversely, the higher affinity and efficacy of APBs at the 5HT2B receptor, which has been conclusively linked to the development of valvular heart disease after prolonged activation, make them less attractive for long term treatment.16 Taken together, the advantages offered by APBs over MDMA demonstrate the potential for gray market designer drugs to offer improvements on current entactogen pharmacotherapies (i.e. MDMA), as research into their safety and efficacy continues in expanded patient populations.
With MDMA proceeding through Phase III clinical trials, a number of pharmaceutical startups focused on psychedelics have taken an interest in novel entactogens. Since the designation of MDMA-assisted therapy as a Breakthrough Therapy by the FDA in 2017, the nonprofit organization MAPS has produced positive results that appear to surpass the efficacy of currently available treatments.17,18 Piggybacking off of MDMA’s recent success, startups including Palo Alto-based Tactogen have begun developing and patenting novel entactogens patterned off of MDMA and analogues such as the APBs.8,19 It should be expected as MDMA moves closer to acceptance as a therapeutic by the FDA that these companies will begin developing related medications to offer physicians a wider range of treatment options and improved safety profiles in the coming years. As this trend continues, the favorable legal status and effect profiles offered by APBs and other novel entactogens will likely position them as attractive candidates for the next generation of entactogenic medicines.

What seems to be missing here are the Beta-ketone of MDMA/MDA.
Mostly known as Methylone, and the APB-analogs of these compounds.
Sasha Shulgin tried to get BK-MDMA registered as an unregistered medicine
but this never worked out.
There are a few MDMA-analogs which could become potential medicines…
…But that is for another discussion.