Note: The concepts discussed below were introduced in Part 1. Read for more background.
Capping off decades of work, Dr. Kuglae Kim of the Department of Pharmacology at the University of North Carolina Chapel Hill (UNCCH) School of Medicine and colleagues have determined the crystal structure of the psychedelic drug LSD bound to the activated 5-HT2A serotonin receptor (5-HT2AR).1 The discovery reported in the journal Cell sheds light on how compounds interact with and activate this canonical mind-altering receptor. The research team was led by Dr. Bryan Roth, a molecular pharmacologist and psychiatrist at the (UNCCH) School of Medicine and director of the National Institute of Mental Health (NIMH) Psychoactive Drug Screening Program (PDSP).
This article takes a deeper dive into what scientists learned from these molecular maps created using X-ray crystallography and cryo-EM, discussed in Part 1. Kim et al.’s findings are ground-breaking, not only for understanding how psychedelics work but also for designing the next generation of neuropsychiatric drugs.
Receptor Binding – LSD’s Unique Interactions with 5-HT2AR
LSD and 25CN-NBOH* act as partial and full agonists for the 5-HT2A receptor (5-HT2AR), respectively, distinguished by the extent of receptor activation.1 The high-resolution structures published by Kim et al. show how each agonist binds to an overlapping pocket within 5-HT2AR (Figure 1). The study reports subtle but critical differences in binding between the two hallucinogens, supporting previous research.
*4-[2-[(2-hydroxyphenyl)methylamino]ethyl]-2,5-dimethoxybenzonitrile
In 2017, Dr. Daniel Wacker and his research team, working with 5-HT2BR, observed that a key amino acid residue (L229ECL2) forms a ‘‘lid’’ over LSD, locking it in place.2 This prolongs LSD’s binding time and, in turn, its biological effects. Now, Kim et al. have found a single residue (S2425.46) unique to 5-HT2AR, which is also necessary for the unusually long action of LSD (Figure 1).1
For comparison, Kim et al. also studied methiothepin, an inverse agonist with opposing receptor effects and vastly different biological activity. Somewhat surprisingly, this compound had a very similar binding position, only slightly expanding the receptor pocket. Interestingly, one key amino acid (D1553.32) was necessary for binding all the compounds to 5-HT2AR.
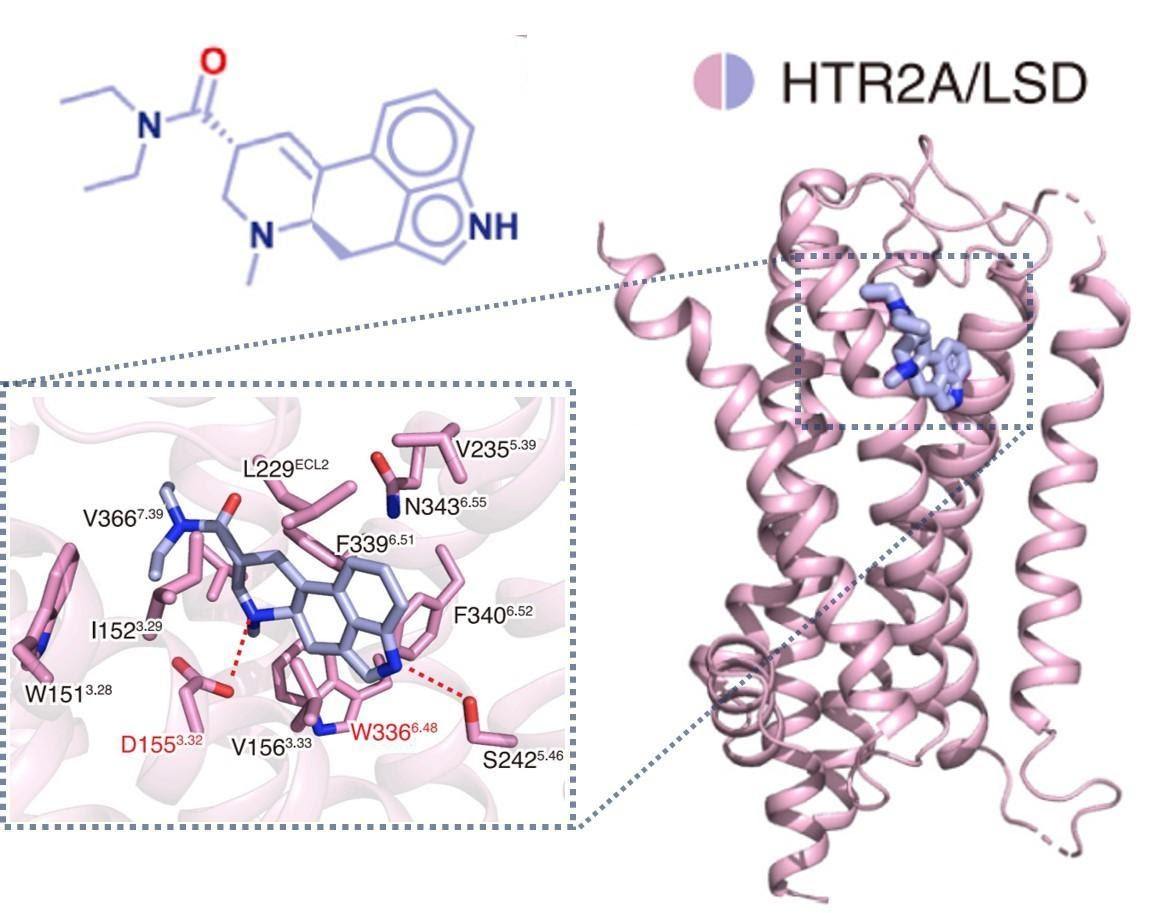
Figure 1: (click to enlarge) The crystal structure of LSD/5-HT2AR.1 The binding pocket is magnified, showing key amino acid residues (identified by their letter code and position within 5-HT2AR) involved in the interaction. (Image compiled from figures in Kim et al. and created by Lily Aleksandrova).
Receptor Activation – LSD Biases Downstream 5-HT2AR Signalling
The structures also revealed important conformational rearrangements that happen during receptor activation. For example, agonist binding displaces a particular 5-HT2AR amino acid, tryptophan W3366.48, which acts as a “molecular switch,’’ turning on the receptor. However, activating the receptor is more complex than just turning it on or off.
Importantly, LSD is also unique in that it is a “biased” agonist. Instead of signalling primarily through the classical G protein-dependent pathway, it preferentially recruits other signalling partners called β-arrestins, with their own set of unique biological effects.
In this study, the drug-receptor complex is seen linked to its G protein (called Gαq) or to β-arrestin (Figure 2). Interestingly, Kim et al. observed that several amino acids within 5-HT2AR are necessary for each of these receptor interactions. For example, a few key amino acid residues underlie 5-HT2AR’s ability to activate Gαq. Curiously, when one is mutated (I18134.51), the receptor binding switches from Gαq to β-arrestin. Such structure-function relationships can begin to explain why an agonist such as LSD might exhibit biased signalling, i.e., a drug stabilized a specific 5-HT2AR signalling complex, ultimately determining its clinical effects.
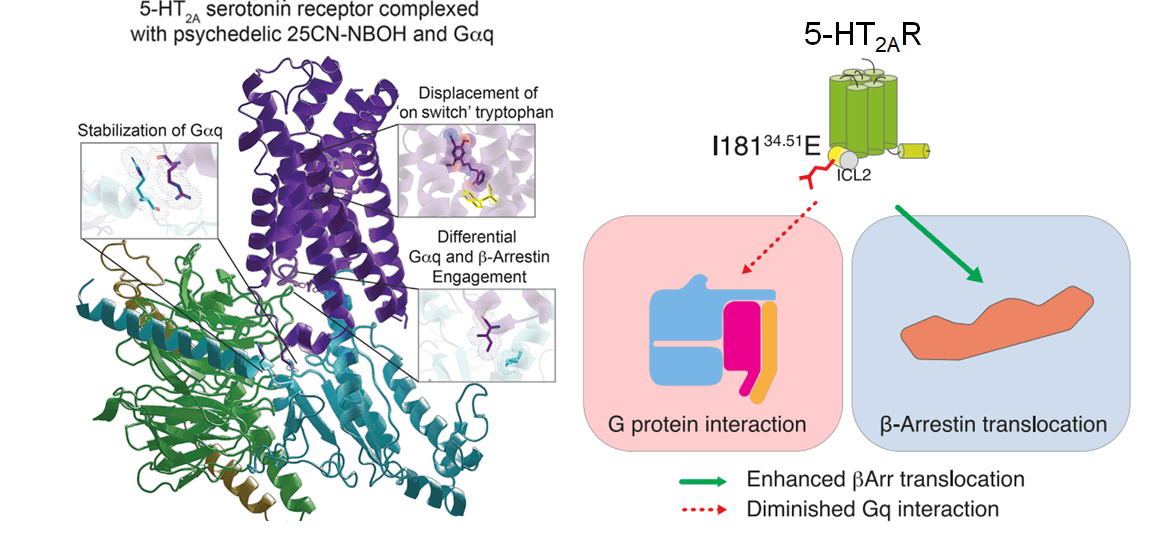
Figure 2: (click to enlarge) The crystal structure of agonist-bound 5-HT2AR linked to Gαq and a model of biased agonism.1 By mutating a key residue within 5-HT2AR (I18134.51) that interacts with Gαq, downstream signalling can be switched to favour β-arrestin (Image compiled from figures in Kim et al. and created by Lily Aleksandrova).
Implications for Neuropsychiatric Drug Discovery
Psychedelics are emerging as potential therapeutics for various neuropsychiatric conditions, including depression, anxiety, and substance abuse. Although crystal structures can never fully explain how a drug works, they improve our mechanistic understanding of its therapeutic and side effects. Such findings help scientists understand the complex relationship between a drug’s molecular structure, its receptor effects and its clinical action.
The study by Kim et al. opens the door to eliminating the hallucinogenic effects of these compounds while retaining their therapeutic effects. IF that’s at all possible. In any case, other researchers are already designing drugs with the anti-inflammatory properties of psychedelics without their hallucinogenic effects. And, Dr. Roth is leading a $26.9 million project for developing better psychiatric medications without major side effects, announced earlier this year.
In their paper, Kim et al. say their work will facilitate “a structure-guided search to identify more selective and efficacious 5-HT2AR agonists as potential innovative neuropsychiatric therapeutics.” Next, the team is developing tools to screen new drug candidates for their ability to activate the receptor without causing hallucinogenic side effects.
In a recent interview with Inverse, Dr. Roth exclaimed,3 “Now we know how psychedelic drugs work – finally!” He added,
By taking apart the signalling processes involved with an LSD experience piece by piece, we might be able to switch some effects on while keeping others off.
