
There are many mysteries surrounding psychedelic mushrooms (aka psilocybin mushrooms or magic mushrooms). One of the most critical questions is very basic; what compounds do these mushrooms contain? Historically, psilocybin (the most abundant compound) has received much of the attention. Psilocybin is a prodrug that the body metabolizes to psilocin, the compound that acts at the serotonin 5-HT2A receptor to produce the psychedelic effect.
Over the years, researchers have identified other compounds in magic mushrooms such as psilocin, baeocystin, norbaeocystin, and aeruginascin. Now, with the renewed interest in the possible therapeutic uses of psychedelics, scientists are revisiting magic mushrooms at a fundamental level—figuring out what compounds are in them.
Study Finds ß-Carboline Compounds in Magic Mushrooms
German scientists have identified new compounds in four species of magic mushrooms and studied their roles in biosynthetic pathways the mushrooms use.1 The researchers isolated the known magic mushroom compounds psilocybin, psilocin, baeocystin, norbaeocystin, and norpsilocin. The new compounds they isolated belong to a family of chemicals known as ß-carbolines.
ß-carbolines are naturally occurring alkaloid compounds.2 They are most commonly known as components of the psychotropic beverage ayahuasca. Examples of ß-carbolines include harmine, harmane, and harmaline (ß-carboline is also another name for the compound norharmane).
Much of the effect of ayahuasca is due to the ability of ß-carbolines to inhibit MAO (monoamine oxidase) enzymes.2,3 This inhibition makes it possible for the psychedelic compound DMT (dimethyltryptamine, another compound in ayahuasca) to pass out of the digestive system and enter the circulation.
ß-carbolines has some psychotropic effects on their own. Still, without their ability to inhibit MAO enzymes, the full effects of ayahuasca are not realized. ß-carbolines also inhibit the uptake of serotonin, dopamine, epinephrine, and norepinephrine via competitive inhibition of the receptors4,5 (Keep in mind that the interactions between an enzyme and a molecule are different from how an allosteric modulator interacts with a molecule).
From a disease standpoint, ß-carbolines are known to play a role in the development of essential tremor (uncontrolled shaking) and have been implicated in Parkinson’s disease.6
Magic Mushrooms Synthesize Several ß-Carbolines
Using 1D and 2D NMR (nuclear magnetic resonance) spectroscopy, Felix Blei and a team of researchers analyzed extracts from Psilocybe cubensis, P. mexicana, P. cyanescens, and P. semilanceata. They identified these ß-carbolines in the extracts: Cordysinin C, Cordysinin D, Harmane, Harmol, Norharmane, and Perlolyrine. Figure 1 shows their chemical structures. These compounds have previously been isolated from fungi and plants in genera such as Cordyceps, Peganum, and Banisteriopsis.7
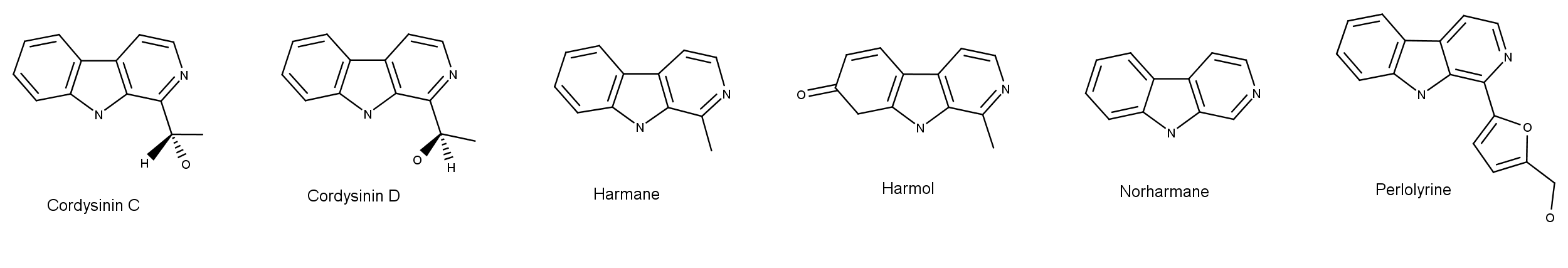
Figure 1: The chemical structures of newly discovered beta-carboline compounds in some species of magic mushrooms1 (click to enlarge).
As part of this study, the researchers used stable-isotope labeling with 13C11-L-tryptophan to show the ß-carbolines were biosynthetic products of the Psilocybe species. This means that ß-carbolines in magic mushrooms may contribute to the entourage effect along with the known compounds. As the researchers put it,
We conclude that Psilocybe mushrooms produce an ayahuasca-like and potentially similarly synergistic set of metabolites that may impact upon onset and duration of their effects. 1
Also, using MALDI-MS (matrix-assisted laser desorption/ionization mass spectroscopy), the researchers showed that the ß-carbolines accumulated at the hyphal apices (the outer edges of the mycelium).
The Effects of Magic Mushrooms vs. Individual Molecules
The Blei et al. paper is a pioneering step in studying the chemistry and variability of the compounds in magic mushrooms. This work highlights the chemical complexity of naturally occurring compounds. It helps with the understanding that ingesting magic mushrooms is very different from taking pure psilocybin (or any other single compound isolated from magic mushrooms). The pharmacology of mushrooms is different than individual compounds and understanding this is critical for optimizing the effects of formulations of magic mushroom compounds.
As is often the case with scientific discovery, these study results answer some questions but also pose more. The presence and known effects of ß-carboline compounds in the Psilocybe species they studied, displays the entourage effect in magic mushrooms in a new light. The study authors sum this up well by saying,
Future pharmacological research is therefore warranted to determine to what extent Psilocybe β-carbolines contribute to the actual psychotropic effects of magic mushrooms.
In a recent interview, Paul Stamets, the world-famous magic mushroom expert told Joe Rogan,
…looking at the natural form of these mushrooms, standardized to psilocybin at a certain concentration versus the pure molecule, I think that is the wave of the future.

I have been waiting for this a long time. So glad these mushrooms are being interrogated more closely. It seems to open a lot of possibilities in observing variations in “potency” with new eyes.
I use them on people who have had strokes.high blood presure.Alzheimer.epilipesy.dementia.ect .they work excelent. Been doing it for 40 years now.
Now i.m learning thier reality
Cheers.
You’ve been giving them to patients for 40 years, and only now you are learning their reality?
Simply True! Tomas!
Excellent Reading.. Thankyou Team
I can’t find the link to the study. Could you maybe add a footer to the article to clearly display the citation?
Never-mind, the footer is below the comment section. It should really go above the comment section.
Very interesting!
Harmala (Syrian Rue) or Banisteropsis Caapi, which are both beta-carboline carriers, harmonize very well with Psilocybin mushrooms as far as the experience goes.
Taken by itself in the right dosage, Harmala or Banisteropsis Caapi has a strong psychotropic effect.
Oooooooof! Be VERY careful suggesting that people use ANY form of MAOI in tandem with psilocybin! This could be EXTREMELY DANGEROUS!
Magic mushrooms are already a carrier of the MAOI beta carbolines, there is NO NEED to add more to the mix as that could seriously harm someone.
There are six references within the article and only two listed below…. I would love to know the other 4 so I can do some deeper investigating!
Whole foods are always better than extracted molecules.
Extremely interesting article. Goes to show how much we still have to learn about Psilocybe Mushrooms. I wonder how this discovery will affect the companies researching a psilocybin treatment using synthesized psilocybin (which would not contain any B-carbolines).
This article appears to imply that magic mushrooms may make tremors worse?
yes, agreed this was a disturbingly cryptic tidbit. To drop that on the table and then not elaborate is unsettling.