
Scientists in Prague, Czechoslovakia, recently identified the compound aeruginascin in Psilocybe cubensis magic mushrooms.1 In their paper published in Drug Testing and Analysis, Klára Gotvaldová and her research team reported: “[a]n extraction procedure and a UHPLC-MS/MS analytical method for the analysis of psilocybin, psilocin, baeocystin, norbaeocystin, and aeruginascin in fungal biomass.” According to the paper, all these analytes were present in the P. cubensis samples studied. The authors noted that “concentrations of aeruginascin are low and have not been reported before.” This paper is the first report of the compound aeruginascin in any mushroom aside from Inocybe aeruginascens2 or Pholiotina cyanopus.3
Coming Up to Speed with Aeruginascin
Aeruginascin was first discovered by Jochen Gartz in 1987 in Inocybe aeruginascens, a psilocybin-containing mushroom.2 Gartz observed that this species had a relatively high concentration of what he hypothesized at the time was aeruginascin. He made another interesting observation in a subsequent 1989 paper.4 Gartz compared and analyzed the experiences from 24 cases of people who accidentally ingested I. aeruginascens to those who accidentally consumed magic mushroom species with high levels of psilocybin and psilocin.
The people ingesting I. aeruginascens reported only euphoric experiences. Gartz described the effects on those consuming mushrooms with high levels of psilocybin and psilocin as an “often slight and in some cases deep dysphoric mood” accompanied by psychosis, panic, and anxiety. Gartz also found in this study that the levels of aeruginascin in the mushrooms were comparable to those of psilocybin and baeocystin.
Over the years, Gartz’s observations have raised questions about whether aeruginascin modifies the clinical effects of psilocybin, psilocin, or both to increase the likelihood of a positive outcome. Gartz himself stated in his 1989 paper the following hypothesis,
Aeruginascin seems to modify the pharmacological action of psilocybin to give an always euphoric mood during the ingestion of mushrooms.
Early this year, Alexander Sherwood and his colleagues synthesized aeruginascin,5 and a research team led by CaaMTech’s Andrew Chadeayne synthesized 4-OH-TMT, the hydrolysis product of aeruginascin and its putative active metabolite.6 In their study, Chadeayne et al. also synthesized 4-AcO-TMT, which serves as a prodrug of 4-OH-TMT and provides an alternative to aeruginascin. Although Sherwood did not study the pharmacology of aeruginascin, Chadeayne et al. demonstrated that 4-OH-TMT binds to 5-HT2A receptors with a greater than expected affinity.
In addition, as Sherwood et al. reviewed in their 2020 paper, there exist anecdotal reports of muscle paralysis, weakness, or both in people after eating some species of psychedelic mushrooms containing aeruginascin.5 Sherwood et al. state, “The speculations on these activities are largely unsubstantiated, though the assumption is tempting given the structural similarity of aeruginascin to bufotenedine, a toad toxin known to be a potent peripherally active 5-HT3 agonist.” The authors added, “…the pharmacological activity of aeruginascin remains unexplored.”
The Impact of This New Study on Psychedelic Mushroom Research
New discoveries add to and change scientific theories all the time. Recall that the magic mushroom compound norpsilocin wasn’t discovered until 20177 and has since been shown to have potency exceeding psilocin’s.5 This study from Gotvaldová et al. highlights the importance of understanding the chemical composition of magic mushrooms–including all the active molecules, not just psilocybin.
Their work indicates that aeruginascin is more prevalent in mushrooms than scientists initially thought. And its presence makes sense given scientists’ understanding of the biosynthetic pathway for making psilocybin and other tryptamine compounds in mushrooms.
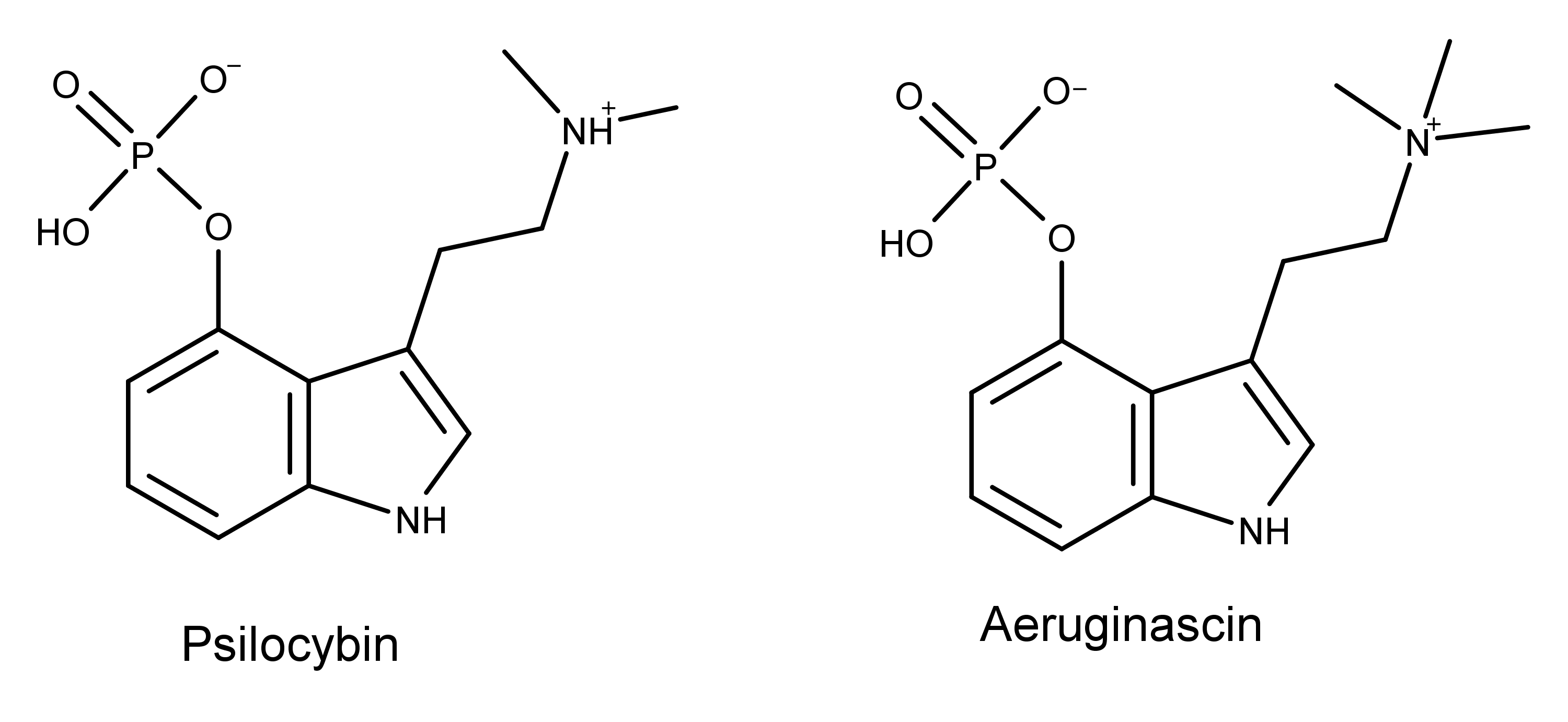
From a structural standpoint, aeruginascin is simply psilocybin with one additional methyl group (see illustration above). Theoretically, another methyl group could be added to psilocybin via the methyltransferase enzymes that mushrooms use to make psilocybin.8 From this perspective, it’s somewhat surprising that aeruginascin isn’t present in more species and varieties of magic mushrooms. Unless it is, and it’s just that no one has gone looking for it yet.

I wonder if this could explain the different affects of various strains of psilocybe mushrooms. e.g. From my personal experience, P. Cubensis (Golden Teacher) has stronger visual and emotional affects than B+, a popular hybrid of P. Cubensis, or possibly P. Azurescens, even when adjusting the B+ dosage up by 25%.
They’re saying it’s found in cubensis in general not different varieties within cubensis, the whole different effects thing fromn different varieties of cubensis is simply placebo. When they say this alkaloid is found in certain mushrooms they mean the whole species not different varieties within. If it’s the same species it’s extremely likely to hold the same amount of active alkaloids, hybrid or not it’s still the same species. Also golden teachers are a mutated variety not found in the wild similar to B+ and Psilocybe azurescens contain the highest levels of alkaloids present in any magic mushroom, if you… Read more »
Saying that the difference from different strains of mushrooms is merely placebo is ridiculous. It’s like saying that every single strain of weed is the same because they all contain THC. You and I both know that so many strains of cannabis exist because there are different effects as a result of all the different compounds in them varying in slight amounts. Many of those compounds and terpenes and new cannabinoids we’ve only discovered in the last few years because of research. In mushrooms they are now discovering new compounds they didn’t realize were there previously because the field of… Read more »
The answer is Trinity cubensis… Lolz… fascinating article…
Extremely important research. Concerning the “… somewhat surprising that aeruginascin isn’t present in more species…” thought, it is well known among Organic Chemists that adding that third methyl group, to produce a quaternary ammonium compound, is much more difficult than adding the first two (to produce a tertiary amine), which is why “quats” are rather expensive even in staple products like surfactants. I believe the same is true with biosyntheses. Even an enzyme is affected by a higher activation energy for a reaction.
Could we genetically modify mushrooms in a lab to make them more medicinal for certain ailments?